Chemical kinetics class 12 notes Most important notes for Revision. Chemical kinetics: It is the branch of chemistry which deals with the study of reaction rates and their mechanisms.
-
- Rate of a reaction: The rate of a reaction can be defined as the change in concentration of a reactant or a product in unit time. For the reaction,
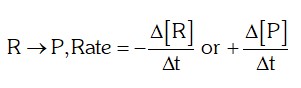
-
- Units of rate: Concentration time–1 i.e., molL–1s–1 or atms–1 for gaseous reactions.
- Average rate of reaction: It is the average value during large time interval.
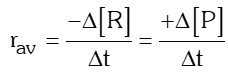
- Instantaneous rate of reaction: It is the rate of a reaction at a particular instant of time i.e., when ∆t approaches zero.
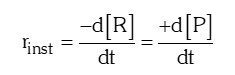
- Factors influencing rate of a reaction: Chemical kinetics class 12 notes
- Concentration: Greater the concentrations of the reactants, faster is the rate of reaction.
- Physical state of reactants: Reactions involving gaseous reactants are faster than reactions containing solid and liquid reactants.
- Temperature: The rate of reaction increases with increase of temperature. For most of the reactions, rate of reaction becomes almost double with 10°C rise of temperature.
- Presence of catalyst: A catalyst generally increases the speed of a reaction.
- Surface are of reactants: For a reaction involving a solid reactant or catalyst, the greater is the surface are, the faster is the reaction.
- Presence of light: Photochemical reactions take place in the presence of light only.
- Activation energy: Lower the activation energy faster in the reaction.
- Rate law and rate constant: The equation that correlates the rate of reaction with concentration of reactants is known as rate law.
- For a simple reaction, A+B→C+D Rate = k[A][B] where k is the rate constant which is equal to the rate of reaction when concentration of each of the reactant is unity.
- Order and molecularity:
Chemical kinetics class 12 notes table

- Half-life of reaction: The time in which the concentration of a reactant is reduced to one half of its initial concentration is called half-life of the reaction.
Rate law, integrated rate law, half-life, units of rate constant and graph for the reactions of different orders: Chemical kinetics class 12 notes

- Pseudo first order reactions: Those reactions which are not truly of the first order but under certain conditions become reactions of the first order are called pseudo first order reactions. e.g.
- Acid hydrolysis of ethyl acetate:
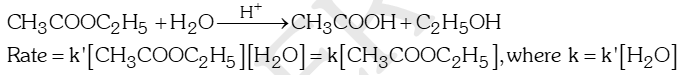
- Acid catalysed inversion of cane sugar:
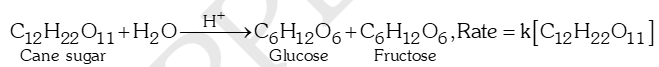
- Effect of temperature on rate of reaction:
- For a chemical reaction with rise in temperature by 10° C, the rate constant is nearly doubled.
- Arrhenius equation:
Chemical kinetics class 12 notes
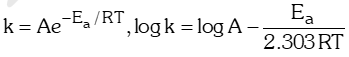
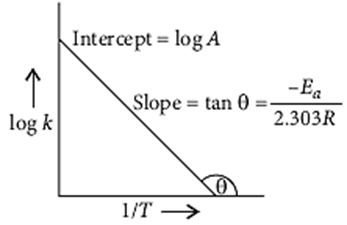
where, k = Rate constant, A = Pre-exponential factor (frequency factor),
Ea Activation energy, T = Temperature,

- Chemical kinetics class 12 notes Most important notes for Revision. Chemical kinetics: It is the branch of chemistry which deals with the study of
- Activation energy: The minimum amount of energy required by reactant molecules to participate in a reaction is called activation energy (Ea).
- Activation energy = Threshold energy – Average kinetic energy of reacting molecules Activation energy ( Ea ) = E(activated complex) –E(ground state) ∆H=Activation energy of forward reaction – Activation energy of backward reaction
Collision theory of chemical reactions:
- Reaction occurs due to collision of molecules.
- All collisions are not effective.
- Effective collisions are those collisions in which molecules collide with sufficient kinetic energy (called threshold energy which is equal to activation energy + energy possessed by reacting species) and proper orientation.
- Rate = PZABe–Ea/RT where, P = probability or steric factor which takes into account the fact that in a collision, molecules must be properly oriented and ZAB = collision frequency (i.e., the no. of collisions per second per unit volume of the reaction mixture) of reactants, A and B.
